Protect the patient you see — and the patient you don’t
PeriWatch Vigilance® is an automated maternal-fetal early warning system for obstetrics that automates the process of identifying patients who may be at risk for adverse outcomes during labor and delivery. Designed to enhance clinical efficiency, standardize care, and facilitate timely intervention, Vigilance is the only AI-based EFM analysis tool commercially available in the United States.

SINGLE PATIENT VIEW
Consolidations of critical data for one patient in a single view with ability to trend 4 or 12 hours. At a glance or by automated notification, clinicians can quickly see if conditions are worsening in each area.
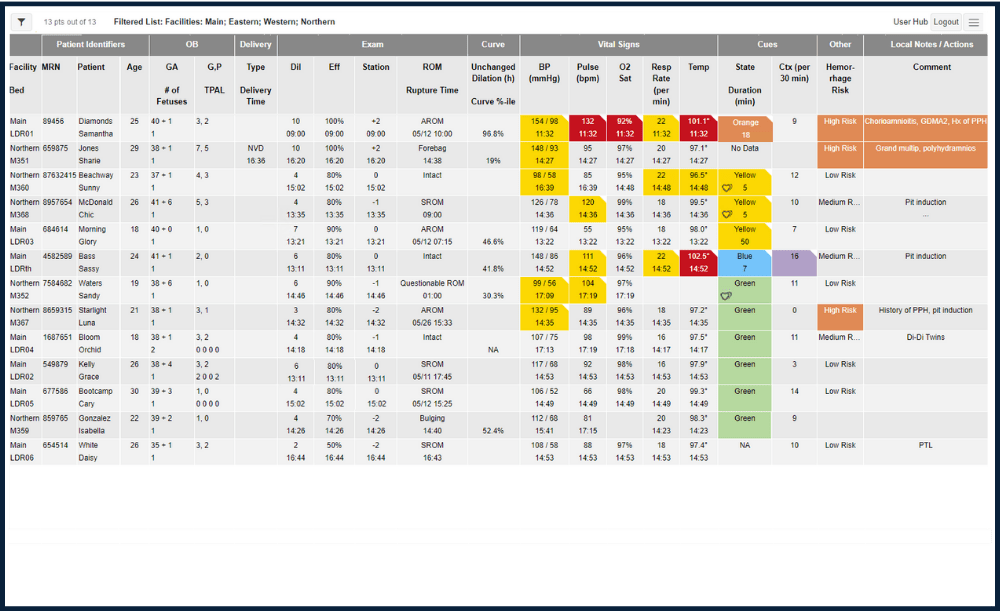
HUB (MULTIPATIENT) VIEW
Live, interactive listing of multiple patients across all or one site, color-coded notifications for quick attention. The list is continuously updated and sorted. One click in a patient row provides access to her detailed personal dashboard.
Cutting-edge Technology for Obstetrics
TIRELESS, TIMELY, CONSISTENT
Always on and analyzing streaming data. Notifies clinicians when maternal vital signs, fetal heart rate, or labor progress exceed safety limits set by the hospital.
MATERNAL-FETAL DYAD
Only automated Early Warning System for Labor and Delivery that considers both mother and fetus. Includes the only EFM analysis tool that has been validated by experts from the NICHD.
WORKS WITH YOUR EXISTING EFM
Works alongside existing EFM systems and EMRs using ADT/HL7 interfacing to avoid costly software replacement and double documentation.
